Anaplasmosis | Vibepedia
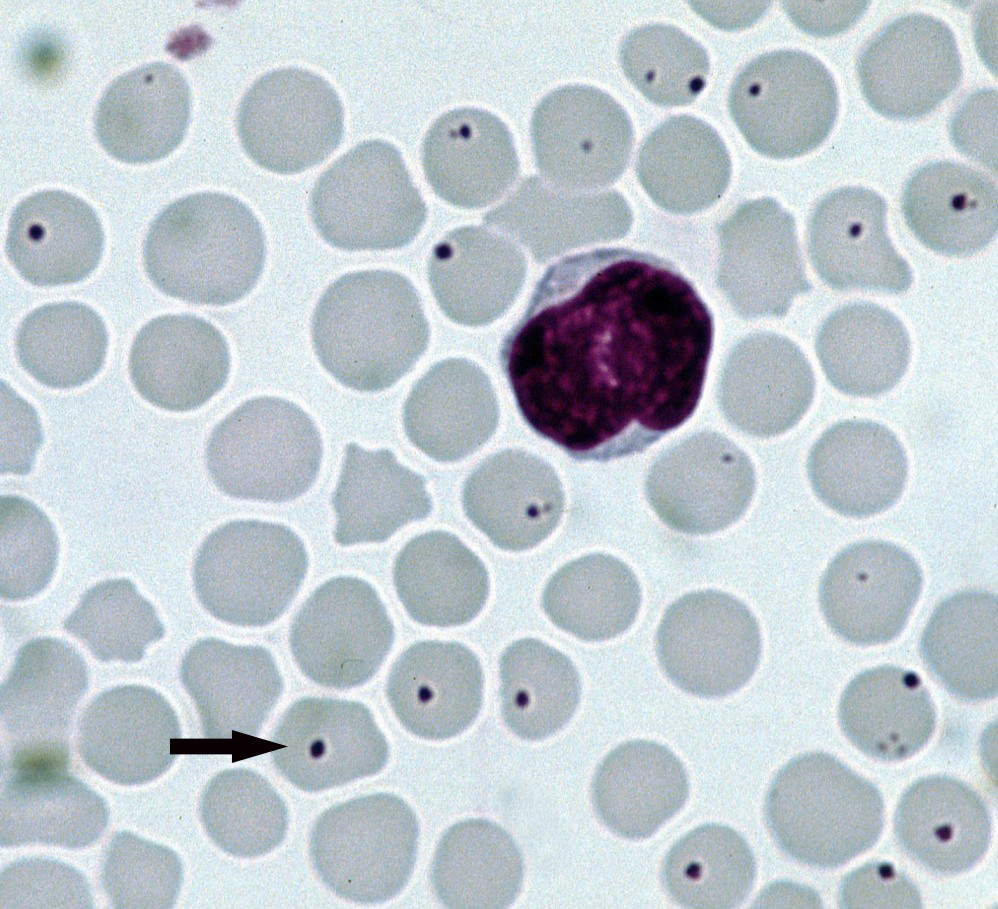
Anaplasmosis is a significant infectious disease primarily affecting livestock, particularly cattle, but also posing risks to dogs, horses, and even humans…
Contents
- 🎵 Origins & History
- ⚙️ How It Works
- 📊 Key Facts & Numbers
- 👥 Key People & Organizations
- 🌍 Cultural Impact & Influence
- ⚡ Current State & Latest Developments
- 🤔 Controversies & Debates
- 🔮 Future Outlook & Predictions
- 💡 Practical Applications
- 📚 Related Topics & Deeper Reading
- Frequently Asked Questions
- References
- Related Topics
Overview
The history of anaplasmosis is intertwined with the development of veterinary medicine and our understanding of [[tick-borne diseases|tick-borne pathogens]]. While the clinical signs in cattle were recognized for centuries, often referred to as "gall sickness" or "yellow bag disease" due to the characteristic jaundice, the causative agent remained elusive. It wasn't until the early 20th century that researchers began to isolate and identify the specific bacteria responsible. The genus Anaplasma was first described in 1910 by Theiler, and Anaplasma marginale, the primary species affecting cattle, was identified shortly thereafter. Early efforts focused on understanding transmission through various tick species, with significant work done by entomologists and veterinarians in regions with high cattle populations, such as North America and Australia. The recognition of Anaplasma phagocytophilum as a distinct pathogen, capable of infecting humans and causing [[human granulocytic anaplasmosis|human granulocytic anaplasmosis (HGA)]], emerged much later, with the first human cases reported in the late 1990s, expanding the scope of anaplasmosis research beyond veterinary concerns.
⚙️ How It Works
Anaplasmosis operates through a sophisticated parasitic invasion of host cells, primarily [[red blood cell|red blood cells]]. The bacteria, such as Anaplasma marginale, adhere to and enter erythrocytes, where they multiply within a vacuole. This intracellular lifestyle shields them from direct immune system attack. However, as the bacteria proliferate, they disrupt the red blood cell's function and integrity. The host's immune system, recognizing the infected cells as foreign, mounts a response. This response involves antibodies and cytotoxic T-cells that target and destroy infected red blood cells. The critical issue is that this immune response is not perfectly targeted; it also leads to the destruction of healthy red blood cells, resulting in [[anemia|hemolytic anemia]]. This anemia is the root cause of many clinical signs, including weakness, pale mucous membranes, and rapid heart rate. Transmission typically occurs when a tick, having fed on an infected animal, subsequently bites a susceptible host, introducing the bacteria into the bloodstream. Mechanical transmission, such as through contaminated surgical instruments or biting flies, can also occur, though it's less common than biological transmission by ticks.
📊 Key Facts & Numbers
Globally, anaplasmosis impacts millions of cattle annually, leading to substantial economic losses estimated in the hundreds of millions of dollars due to reduced milk production, weight loss, decreased fertility, and animal mortality. In the United States alone, [[anaplasma-marginale|Anaplasma marginale]] infections in cattle are estimated to cause over $300 million in losses each year. Anaplasma phagocytophilum is responsible for [[human granulocytic anaplasmosis|human granulocytic anaplasmosis (HGA)]], a disease that saw approximately 3,000 reported cases in the U.S. in 2022, according to the [[Centers for Disease Control and Prevention|CDC]]. Canine anaplasmosis, caused by Anaplasma platys and Anaplasma phagocytophilum, is also prevalent, with seroprevalence rates varying widely by region, sometimes exceeding 20% in certain dog populations. The disease is endemic in many parts of the world, with a particular prevalence in tropical and subtropical regions where tick populations are robust year-round. The economic burden extends beyond direct losses, encompassing costs for diagnosis, treatment, and tick control measures, which can run into millions of dollars annually for livestock industries.
👥 Key People & Organizations
The scientific community dedicated to understanding and combating anaplasmosis is vast and multidisciplinary. Key figures in early veterinary pathology, such as [[Sir Arnold Theiler|Sir Arnold Theiler]], laid foundational groundwork for identifying tick-borne diseases in livestock. More recently, researchers like Dr. Gary A. Splitter at the [[University of Wisconsin-Madison|University of Wisconsin-Madison]] have made significant contributions to understanding the molecular mechanisms of Anaplasma phagocytophilum infection and host immune responses. Organizations like the [[Centers for Disease Control and Prevention|CDC]] in the United States play a crucial role in monitoring human cases and providing public health guidance. Veterinary diagnostic laboratories worldwide, including those affiliated with agricultural universities and government agencies like the [[United States Department of Agriculture|USDA]], are vital for disease surveillance and diagnosis. The [[World Organisation for Animal Health|OIE]] (World Organisation for Animal Health) also tracks anaplasmosis as a significant animal disease, promoting international standards for diagnosis and control.
🌍 Cultural Impact & Influence
Anaplasmosis has a subtle yet pervasive cultural footprint, primarily within agricultural communities and veterinary circles. The colloquial names like "yellow bag" or "gall sickness" are embedded in the vernacular of cattle farmers, reflecting generations of experience with the disease. In veterinary education, anaplasmosis serves as a classic case study for understanding [[hemolytic anemia|hemolytic anemia]] and the complexities of [[vector-borne diseases|vector-borne transmission]]. The emergence of human anaplasmosis has also brought the disease into broader public health awareness, albeit often overshadowed by more widely known tick-borne illnesses like [[Lyme disease|Lyme disease]]. The visual symptom of jaundice in infected animals has also found its way into anecdotal descriptions and historical agricultural texts, contributing to a shared understanding of animal health challenges within farming cultures. The ongoing battle against anaplasmosis underscores humanity's continuous effort to coexist with and manage the natural world, particularly the intricate relationships between pathogens, vectors, and hosts.
⚡ Current State & Latest Developments
The current landscape of anaplasmosis research and control is dynamic. Recent advancements in [[genomics|genomic sequencing]] have allowed for more precise identification of bacterial strains and their geographic distribution, aiding in targeted control strategies. The development of new diagnostic tools, including [[polymerase chain reaction|PCR]]-based assays and improved serological tests, offers faster and more accurate detection of infections in both animals and humans. In veterinary medicine, there's a growing emphasis on integrated tick management programs, combining chemical control with environmental management and host resistance strategies. For human anaplasmosis, public health efforts focus on raising awareness among clinicians and the public about the disease's symptoms and the importance of tick bite prevention, especially in endemic areas. Research is also ongoing into potential [[vaccine|vaccine]] development for both animal and human anaplasmosis, though challenges remain due to the bacteria's complex lifecycle and immune evasion strategies. The increasing global trade and travel also raise concerns about the potential spread of anaplasmosis to new regions.
🤔 Controversies & Debates
One of the persistent controversies surrounding anaplasmosis revolves around the efficacy and long-term impact of certain treatment protocols, particularly the widespread use of [[tetracycline|tetracyclines]] in livestock. While effective in reducing clinical signs and bacterial load, concerns exist about the development of antibiotic resistance and the potential for residual bacterial persistence in treated animals, which can continue to serve as reservoirs for infection. Another debate centers on the economic feasibility and environmental impact of aggressive tick eradication programs versus integrated pest management strategies. For human anaplasmosis, there's ongoing discussion about underdiagnosis and the need for greater clinician awareness, as symptoms can mimic other febrile illnesses, leading to delayed treatment. The zoonotic potential of certain Anaplasma species, while recognized, is still an area where precise risk assessment and public health messaging are being refined, balancing alarm with accurate information.
🔮 Future Outlook & Predictions
The future of anaplasmosis management likely lies in a multi-pronged approach combining advanced diagnostics, novel therapeutics, and robust prevention strategies. Researchers are exploring alternative treatment options beyond traditional antibiotics, including [[antimicrobial peptides|antimicrobial peptides]] and phage therapy, to combat potential antibiotic resistance. The development of more effective and safer [[vaccine|vaccines]] for both livestock and potentially humans remains a high priority, though significant hurdles exist in eliciting a protective immune response against these intracellular pathogens. Enhanced surveillance systems, leveraging [[big data|big data]] analytics and [[artificial intelligence|AI]] for predictive modeling of tick populations and disease outbreaks, will become increasingly crucial. Furthermore, a greater understanding of the host-pathogen interactions at a molecular level could unlock new targets for therapeutic intervention. Public health campaigns focused on tick bite prevention and early symptom recognition will continue to be vital for mitigating human cases, especially as climate change potentially alters tick distribution and activity patterns.
💡 Practical Applications
In veterinary medicine, the primary application of knowledge about anaplasmosis is in disease prevention and treatment. This includes implementing strict [[tick control|tick control]] measures on farms, such as pasture management and the use of acaricides, alongside regular diagnostic testing of livestock. Treatment of infected animals typically involves [[antibiotic therapy|antibiotic therapy]], most commonly with tetracyclines, to reduce clinical signs and mortality. For canine anaplasmosis, similar diagnostic and therapeutic approaches are employed. In human health, the practical application focuses on [[public health|public health]] education regarding tick bite prevention—using repellents, wearing protective clothing, and performing tick checks—and prompt medical attention if symptoms arise. Early diagnosis and treatment of human anaplasmosis with antibiotics like doxycycline are crucial for preventing severe complications. Research into anaplasmosis also informs broader efforts in [[zoonotic disease|zoonotic disease]] surveillance and the development of diagnostic platforms applicable to other tick-borne pathogens.
Key Facts
- Year
- Early 20th Century (identification of causative agent)
- Origin
- Global (primarily tropical and subtropical regions)
- Category
- science
- Type
- topic
Frequently Asked Questions
What are the main symptoms of anaplasmosis in cattle?
In cattle, anaplasmosis, often called "yellow bag" or "gall sickness," typically presents with high fever, loss of appetite, weight loss, and decreased milk production. A hallmark sign is anemia, leading to pale mucous membranes and weakness. Jaundice, a yellowing of the skin and eyes, is also common, hence the "yellow bag" moniker, referring to the yellowish color of the udder. Some animals may also exhibit diarrhea and neurological signs. The severity can range from mild, subclinical infections to severe, life-threatening cases, particularly in adult cattle.
How is anaplasmosis transmitted to humans?
Anaplasmosis is transmitted to humans primarily through the bite of infected ticks, most commonly the blacklegged tick (Ixodes scapularis) and the western blacklegged tick (Ixodes pacificus) in North America. These are the same ticks that transmit [[Lyme disease|Lyme disease]] and [[Ehrlichiosis|ehrlichiosis]]. The bacteria, Anaplasma phagocytophilum, infect white blood cells called granulocytes. While less common than tick transmission, mechanical transmission through contaminated blood transfusions or organ transplants has also been documented, highlighting the importance of screening blood donors in endemic areas.
What is the economic impact of anaplasmosis on the livestock industry?
The economic impact of anaplasmosis on the livestock industry is substantial, running into hundreds of millions of dollars annually worldwide. In cattle, losses stem from reduced milk yield, decreased weight gain, infertility, and increased mortality rates. The cost of diagnosis, treatment with antibiotics like tetracyclines, and implementing tick control measures further adds to the financial burden. In regions where anaplasmosis is endemic, it can significantly affect the profitability and sustainability of cattle farming operations, prompting significant investment in disease management and prevention strategies by producers and governments alike.
Can anaplasmosis be prevented in animals?
Prevention of anaplasmosis in animals focuses on reducing exposure to infected ticks and managing the spread of the disease. This includes implementing rigorous [[tick control|tick control]] programs on farms, such as using acaricides on livestock and in pastures, and managing vegetation to reduce tick habitats. For cattle, vaccination strategies are being explored, though current vaccines have limitations. Maintaining good animal health and nutrition can also help animals better withstand potential infections. In areas where anaplasmosis is prevalent, regular diagnostic testing and prompt isolation and treatment of infected animals are crucial to prevent further spread within the herd. Careful management of animal movement and quarantine procedures for new arrivals can also mitigate risk.
Why is anaplasmosis sometimes called 'yellow fever' or 'yellow bag'?
The names "yellow fever" and "yellow bag" are colloquial terms used to describe anaplasmosis, particularly in cattle, due to a prominent clinical sign: jaundice. Jaundice is a yellowing of the skin, mucous membranes, and sclera (the white part of the eyes) caused by a buildup of bilirubin in the blood. This occurs because the bacteria infect and destroy red blood cells, leading to severe anemia. As the body breaks down these red blood cells, bilirubin is produced, and in anaplasmosis, the liver's ability to process this bilirubin is overwhelmed, resulting in the characteristic yellow discoloration. The term "yellow bag" specifically refers to the yellowish appearance of the udder in infected dairy cows.
How is anaplasmosis diagnosed in humans and animals?
Diagnosis of anaplasmosis relies on a combination of clinical signs, epidemiological data, and laboratory tests. In animals, veterinarians may suspect anaplasmosis based on symptoms like fever and anemia, and confirm it through blood smears examined under a microscope, serological tests (like ELISA) to detect antibodies, or molecular tests (like [[polymerase chain reaction|PCR]]) to detect bacterial DNA. For humans, diagnosis involves similar laboratory methods, with PCR being highly sensitive for detecting active infection and serology used to identify past exposure. Blood smears can also reveal the characteristic intracellular morulae (clusters of bacteria) within white blood cells, particularly in cases of human granulocytic anaplasmosis.
What are the long-term consequences of anaplasmosis infection?
In animals, long-term consequences of anaplasmosis can include chronic anemia, reduced productivity (lower milk yield, slower weight gain), and decreased reproductive efficiency, leading to economic losses for farmers. Some animals may become carriers, remaining infected and potentially serving as reservoirs for transmission even after clinical signs resolve. In humans, while most cases resolve with antibiotic treatment, severe infections can lead to complications such as [[respiratory failure|respiratory failure]], [[kidney failure|kidney failure]], [[neurological complications|neurological complications]], and in rare instances, death. Chronic or relapsing symptoms can occur, and some individuals may experience prolonged fatigue and joint pain following infection, similar to post-viral syndromes.